Working Hour: 08.00am - 09.00pm
Email: info@anbh.co.in
Core Verticals
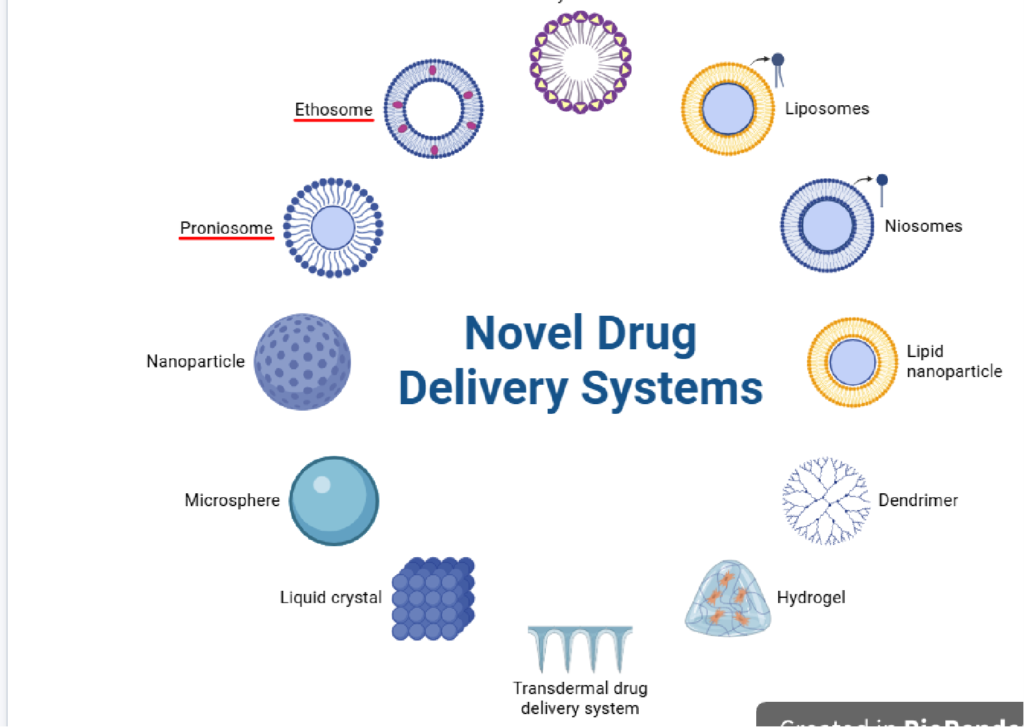

1. INTRODUCTION
What is known? (our understanding)
2. LITERATURE REVIEW
what’s the gap we want to fill?
3. RATIONAL
Why and how should we fill the gap?
4. METHODS
What did you do
5. RESULT AND DISCUSSION
What results did you get ? How do the results fill the gap?
6. CONCLUSION
What does this mean for us going forward?
7. SUMMARY
8. REFERENCES
Where have you collected the information?

Pre-formulation/compatibility studies
Nanoformulation development by QbD approach
Optimization
Characterization
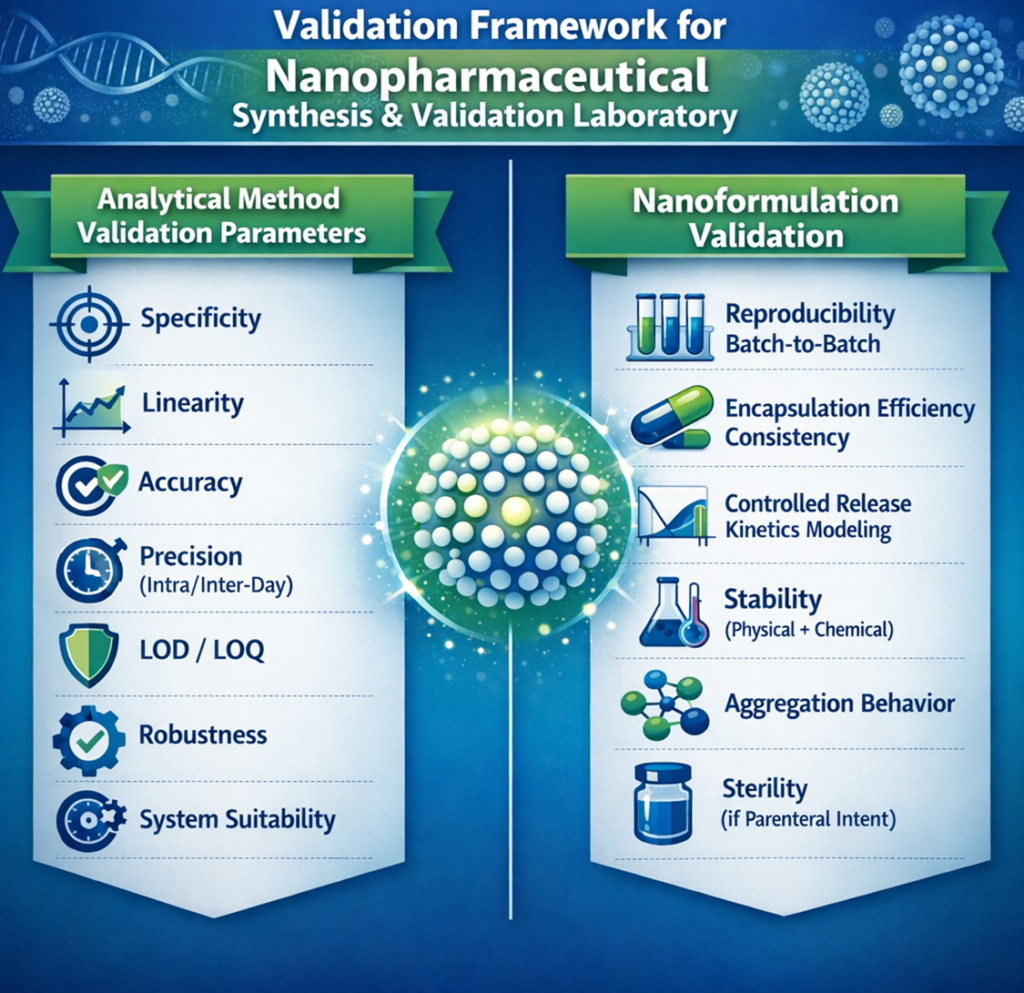
Optimized formulation validation
In-vitro Release profile
Reproducibility by Pilot studies
Reproducibility by Batch to Batch
Encapsulation efficiency consistency
Controlled Release modelling
Stability
Aggregation behaviour
Packaging
Screening of the column and mobile phase on the basis of the drug molecule properties Method development at different ratios of the mobile phase on the single drug/combination drug
Optimization of the developing method via QbD Approach
Validation of the optimized method:
Specificity
Linearity
Accuracy
Precision
LOD/LOQ
Robustness
System suitability